Company Overview
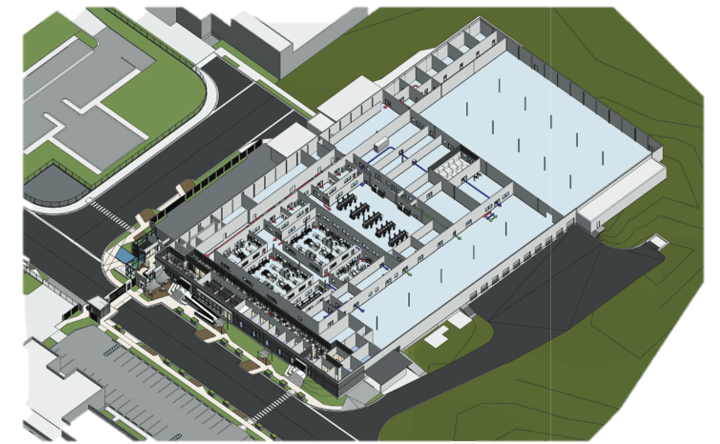
OcyonBio provides dedicated autonomous manufacturing capacity with interconnected infrastructure and systems to support phased appropriate development for early development, pre-clinical, clinical, and commercial start. We behave more like a cGMP incubator space with all regulatory, systems capabilities, and resources to enable CMC data to support regulatory applications. Manufacturing and development spaces are designed to be autonomous while being interconnected to systems required to support clinical and commercial requirements.
OcyonBio provides a company with its own space, so there is no need to build an expensive facility. Providing flexibility to protect IP, manage schedules, resources, and new product introduction reducing overall cost and risks.